Key Highlights
- SAP-related compliance errors are greatly decreased by practical training and real-time user support.
- A scalable governance framework is essential for multinational pharmaceutical companies that operate across borders.
- Keeping an eye on adoption metrics helps spot gaps early and avoids problems with compliance.
- Standardized procedures, role-based training, and ongoing oversight are all components of effective governance that manage SAP usage.
Introduction
Adopting SAP in the pharmaceutical sector is a compliance-critical undertaking that goes much beyond a digital transformation project. Strict international regulations, including GxP, FDA 21 CFR Part 11, and EMA guidelines, must be followed in every transaction, procedure, and data input. Pharma must make sure that adoption is controlled, validated, and auditable at every stage, in contrast to other businesses where system adoption is primarily motivated by efficiency.
An SAP adoption governance framework is crucial in this situation. Organizations run the risk of uneven system utilization, audit failures, and expensive compliance violations in the absence of a systematic approach. Global teams, numerous SAP modules, and changing regulatory requirements all add to the complexity.
While maintaining business agility, a clear governance architecture guarantees that SAP deployment is standardized, traceable, and compliant. But governance is more than simply policies; user support and training play a major role.
This is where tools like Assima Train play an important role, allowing businesses to provide SAP training that is compliant, scalable, simulation-based, and entirely compliant with governance regulations.
Why SAP Adoption in the Pharmaceutical Industry Depends on Governance?
Pharmaceutical firms are subject to some of the world’s most stringent regulations. All systems that affect patient safety and product quality must be validated, managed, and auditable according to frameworks like FDA rules, EMA standards, and GxP (Good Practice recommendations). These rules immediately apply to SAP systems, which are frequently the foundation of operations, including manufacturing, supply chain, and quality control. Governance is therefore unavoidable. Organizations run significant risks if they don’t have a robust SAP governance framework:
Compliance failures: Inconsistent processes can violate regulatory requirements.
Audit risks: Lack of traceability and documentation can lead to failed inspections.
Data inconsistencies: Poor adoption leads to inaccurate or incomplete data, affecting decision-making and product integrity.
According to recent industry reports, a large percentage of operational risks in pharmaceutical IT systems are tied to compliance. In SAP, even small user mistakes can lead to significant violations of compliance. Governance guarantees that all procedures are appropriately recorded, users are held responsible for their activities, and SAP usage is consistent across departments. It is a regulatory necessity in a regulated environment.
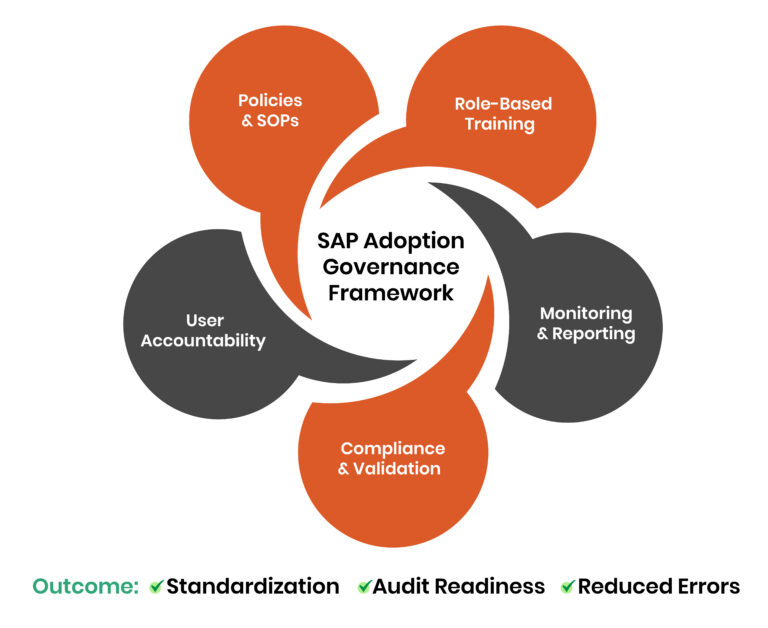
What does a SAP Adoption Governance Framework mean?
An organized method that outlines how a company oversees, regulates, and maximizes the usage of its SAP systems is called a SAP adoption governance framework. It ensures that users interact with SAP in a way that is consistent, compliant, and aligned with business objectives. Fundamentally, the framework concentrates on three main results:
- Standardization: Ensuring that all users follow predefined workflows and procedures.
- Compliance: Aligning system usage with regulatory requirements and validation standards.
- User accountability: Tracking and managing user actions to ensure responsibility and traceability.
Typically, a strong governance structure consists of:
Policies and Procedures
Standard Operating Procedures (SOPs) that are well-defined and in line with SAP workflows guarantee that users carry out duties accurately and consistently.
Training Programs
Role-based, compliance-focused training guarantees that users comprehend not only how to use SAP but also why certain procedures must be followed.
Monitoring & Control Mechanisms
Organizations could recognize deviations early and keep control by using real-time monitoring of user behavior, audit logs, and performance metrics.
Going beyond IT, governance becomes a cross-functional discipline including quality, compliance, and operations teams in regulated pharmaceutical environments. Without this strategy, SAP adoption becomes risky and dispersed.

Key Challenges in SAP Adoption for Regulated Pharma
Compliance & Validation
Validation is one of the main obstacles to pharmaceutical SAP implementation. To comply with regulations, each SAP process needs to be tested, approved, and recorded. This slows down system updates and adds to the load of documentation.
Agility is challenging because even minor updates need to be re-validated. Organizations need to strike a balance between stringent compliance rules and innovation.
Complex Systems & Workflows
Pharmaceutical SAP environments are very complicated, involving several modules like SAP S/4HANA, SAP QM (Quality Management), and SAP EWM (Extended Warehouse Management). Because these systems are interdependent, a modification in one area may have an effect on other processes.
Cross-functional dependencies increase the complexity even further. For instance, a manufacturing modification may have concurrent effects on supply chain, regulatory reporting, and quality.
User Adoption
User acceptance is still a constant problem even with strong systems. Some common issues include:
- Resistance to change, especially among experienced employees
- Lack of contextual, real-time support
- Over-reliance on manual workarounds
According to studies, adoption issues affect up to 70% of digital transformation projects, and the pharmaceutical industry is no exception. Poor adoption becomes a compliance risk in regulated situations rather than just an efficiency issue. Users who divert from validated procedures can end up subject to audit results and even legal repercussions.
Essential Components of an Effective SAP Governance Framework
It takes a combination of process discipline, user empowerment, and ongoing monitoring to create an efficient SAP adoption governance system.
Standardized Procedures
Governance is built on standardization. SOPs that are in line with SAP workflows and legal requirements must be defined by organizations. All agencies and areas should have uniform enforcement of these protocols, which should be transparent and easily accessible.
Role-Based Training
Every user interacts with SAP differently. The needs of a manufacturing or quality assurance user are very different from those of a financial user. Role-based training guarantees that every user gets education that is pertinent and focused. More significantly, by emphasizing the unique duties and dangers connected to each position, it strengthens compliance.
Monitoring & Reporting
Visibility is essential to governance. Businesses need to keep tabs on:
- User activity
- Process adherence
- Error rates
In addition to highlighting areas that require further training or process modifications, advanced reporting systems aid in identifying adoption gaps.
Audit Readiness
Audits are a given in the pharmaceutical industry. Organizations that have a robust governance system are always prepared for audits by upholding:
- Complete records
- The capacity to track user behaviors
- Training and compliance evidence
Maintaining audit readiness is a continual process that requires constant effort. When combined, these components provide a regulated environment where SAP implementation is both effective and completely compliant.
How Does Assima Train Help Pharma's SAP Governance?
Assima Train stands out as a potent solution for regulated pharmaceutical environments because technology is essential to enabling governance.
Simulation-Based Training for Compliance
Users can practice SAP procedures in a simulated environment that replicates the real system using Assima Train. As a result, users can grow and make mistakes without affecting production data. This is crucial for companies that rely heavily on compliance. It guarantees that users are ready to engage with verified systems.
Consistent Global Training Delivery
Pharmaceutical companies frequently operate in several different locations, each with unique regulatory requirements. Assima Train permits localization when necessary while enabling uniform training delivery across regions. This guarantees uniformity without compromising adaptability.
Error Reduction & Support
Assima dramatically lowers user errors by training users in real-world circumstances, which is one of the main causes of compliance problems. The technology also offers proof of training and paperwork, enabling audit requirements and proving compliance.
Scalable Training Framework
Training requirements develop quickly as businesses expand or add new SAP modules. Assima Train offers a scalable architecture that can accommodate thousands of users without sacrificing quality. Assima Train essentially ensures that policies are not only created but also successfully implemented by bridging the gap between governance and implementation.
Conclusion
SAP adoption cannot be left up to chance in regulated pharmaceutical workplaces. It necessitates an organized, clear governance architecture that guarantees responsibility, uniformity, and compliance at all levels. Training is the key to successful adoption, even though procedures and regulations serve as the cornerstone. Even the strongest governance structures might fail if users are not adequately taught.
Here, Assima Train helps businesses to provide SAP training that is scalable, compliant, and successful while also smoothly integrating with governance goals. In the end, a robust SAP adoption governance structure is about creating an organization that is resilient, effective, and prepared for the future rather than just fulfilling legal obligations.
Explore Assima Train today to strengthen software adoption across your organisation.
Frequently Asked Questions
Let’s Answer Some of Your Questions.
Assima Train offers scalable, simulation-based training that enhances user competency, lowers errors, and satisfies audit and compliance requirements.
